|
|
More from Dr. Mayer on Carbon! Part 1 of 2
11/11/2010
| Lawrence Mayer, University of Maine
(Ocean Systems)
Tags: 10.06.10 Webinar, carbon cycle, scientist post
|
 Dr. Larry Mayer presents a concept map at a COSEE-OS workshop in February of 2010. |
Further, this webinar was designed for a target audience likely to consist of science teachers rather than high school students. That new audience implied changing the concept map from one emphasizing content to one emphasizing conceptual tools. I hope that science teachers will find the carbon cycle both inherently interesting and, more importantly, a great vehicle to teach students fundamental tools of quantitative thinking. I therefore placed more stress on the identification/ inventory/ flux/ residence time sequence of ideas than for the original high school student audience. Teachers can export these ideas about quantification to a wide range of topics in addition to the carbon cycle.
Note: Many thanks to the educators who submitted some really interesting questions in the pre-webinar survey. Larry has taken the time to let us “pick his brain” and answered some of his favorite questions below.
Has the abundance of carbon in the ocean basins increased significantly in the last 50 years? How has the Gulf oil spill changed this?
On the order of half of the carbon dioxide that has been released into the atmosphere through the burning of fossil fuels has been absorbed by the ocean. That’s a big fraction of fossil fuel emissions, but it makes only a small dent in concentration of carbon in the ocean. A bit more worrisome, however, is the fact that this added carbon lowers the pH of the ocean water, which can lead to other problems. The Gulf oil spill added carbon in a visually spectacular way, with significant impacts on some biota, but again it didn’t make a big difference in the overall carbon content of ocean water. Just like the acidic carbon coming from the atmosphere, it creates impacts quite out of proportion to its simple concentration. That’s true of most pollutants.
To what extent do volcanoes and forest fires contribute to carbon cycles and deposition?
The very long term carbon cycle (billions of years) is strongly influenced by volcanoes, because most carbon originally came from the deep earth to the surface via volcanoes. On the shorter time scales that we are concerned with, volcanoes can affect global temperature by putting up an ash “curtain” that leads to cooling of the earth and reduction in plant production. The ash from volcanoes might be involved in affecting the carbon chemistry of water, though likely on longer time scales than humans need to worry about. Forest fires contribute tree carbon directly to the atmosphere, though this contribution is not a major one. There is emerging research to suggest that the soot from fires is important in sequestering carbon in soils and maybe sediments as well.
Is there a relationship between the carbon cycle and water cycle in atmosphere?
There are lots of relationships, both direct and indirect. Water carries carbon, so water movement often determines where carbon goes. Carbon dissolved in water can make it more acidic, so that weathering rocks depends in part on this chemical reaction. Both carbon dioxide and water vapor are greenhouse gases, so that their respective influences on temperature will affect one another. Clouds affect light, and therefore how much phytoplankton can be produced (see above). Gee, you could make a neat concept map of these relationships!
Sounds like Larry has issued a ‘concept map challenge’ – show us what you and your students create!
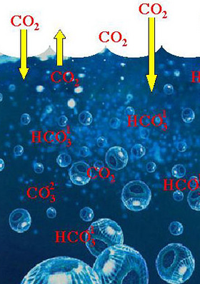 Carbon dioxide can be absorbed by the ocean and used by plankton. |
An intense zone of food web reworking is located just below the upper ocean layer where there’s enough light for phytoplankton; some scientists like to call these depths the “Twilight Zone”. Thus, the settling carbon flux to the deep sea depends on the success of this carbon in transiting through the Twilight Zone.
I'd like a better understanding of the relationships among wave action, ocean acidity, light penetration into the ocean, and carbon sequestration. Also, how do light and currents play a role?
One of the limiting mechanisms for oceanic uptake of carbon from the atmosphere is simply mixing it in - across the air-to-water interface. Just like you can cool hot tea by blowing on it, you can dissolve more atmospheric carbon dioxide into the ocean’s surface by increasing wind speed. Computer models and hence estimates of oceanic uptake generally involve a wind speed component. Waves of course are a symptom of wind, and promote mixing by burying bubbles of the atmosphere into the water column. Besides simple mixing, this burial takes gas bubbles to zones of higher pressure, where the gases are more soluble. For example, gases are twice as soluble at a depth of 10 meters than at the surface. Currents can have similar effects, but are not as effective as wave mixing at the sea surface.
Light is important because it limits phytoplankton growth, but don’t forget their need for “miracle grow” – i.e. nutrients. Essential nutrients for phytoplankton such as nitrate and phosphate are more plentiful at deeper depths, but of course sunlight is most accessible closer to the ocean surface. Anytime you can mix nutrients upward into the water column, or transmit more light downward into the water column, you enlarge the “sweet spot” where phytoplankton can get both and thrive.
Stay tuned for even more answers from Dr. Mayer coming soon!
Have thoughts on this post? Send us a note and we'll share your ideas!
|

