|
|
More from Dr. Mayer on Carbon! Part 2 of 2
11/29/2010
| Lawrence Mayer, University of Maine
(Ocean Systems)
Tags: 10.06.10 Webinar, carbon cycle, scientist post, carbon sequestration
|
In this second installment, Dr. Larry Mayer (University of Maine) answers some additional questions posed by webinar participants. To watch Dr. Mayer's webinar presentation, you can visit the archive page.
You talked a lot in your webinar about carbon being sequestered in ocean sediments. How do scientists determine the length of time that carbon will be sequestered for?
One straightforward way is to use the residence time concept as explained in the webinar (e.g., iPod song list residence times). We measure how much carbon is in a particular pool (e.g., concentration of carbon dioxide in the atmosphere). Then we find the rate at which it enters and leaves that pool. Using the atmosphere example, we would add up carbon dioxide inputs to the atmosphere from all the sources we can find (e.g, organisms, volcanoes, fossil fuel usage, etc.). We would then divide the two terms to get a residence time, and use that as an estimate of sequestration time.
The actual measurements for this determination depend on which pool or flux is being measured. Let’s look at a couple of examples. People are starting to put towers with carbon dioxide sensors over various ecosystems, such as forests or marshes. By continually measuring both the concentration of carbon dioxide and the direction of air flow from these towers, they can calculate the rate of carbon dioxide movement upward or downward. Think of it as measuring the number of box cars going by a train crossing (over a certain period of time) multiplied by the amount of goodies loaded in each box car. The resulting calculation would be an estimate of the “flux of goodies” going past the crossing per time period. In the webinar, we also mentioned the use of sediment traps to measure settling rates of carbon-rich particles as they sink through the water column towards the ocean bottom. These devices work just like a rain gauge. Last, we can assess the rate of carbon burial in sediments by combining measurements of total sediment burial (how long did it take for that layer to get deposited?) with the carbon concentration in the sediment.
What practical artificial means of carbon sequestration are being investigated?
There are lots of proposals on the table, ranging from recovery of carbon dioxide from power plant exhausts and injection into rocks or the deep ocean, reestablishing forests to build up living carbon stocks in tree trunks and soil, changing soil management in fields to increase their organic matter content, and many others.
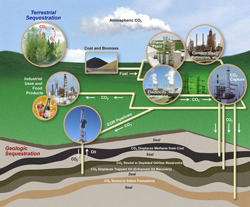 Several carbon sequestration methods. (Provided by the US Dept. of Energy). Click for a larger view. |
Is artificial carbon sequestration feasible -considering that this carbon would need to be buried in sediment else it would be cycled back up into the thermohaline circulation in 1000 years?
While the deep water injection technologies being studied do suffer from this “1000 year threat”, many people regard the possibility as still useful because it “kicks the can down the road” to a time when some future technology may be able to solve that problem.
Which is more efficient: pumping carbon underwater, or into the deep spent oil wells in Texas and elsewhere?
I’m not sure which is more efficient, and of course efficiency depends on what you are comparing with what (for example, have you accounted for the entire life cycle of each process?). Exploring the web sites above might provide some insight. If efficiency can be assessed via costs (a great way to sum up lots of information), then many of these numbers still need to be worked out.
|

